RNI advises you on regulatory and scientific news and organises and participates in private events to inform manufacturers.
Latests news

What’s new in Nutrition & Food Supplements ?

Nitrosamines – White paper written by Manolina Kontaxaki

Micronutrients health related guidance values: what’s new for vitamin B6, selenium and copper?

USA – New Guidance for Advertising Claims of Supplements

RULE 21 – New Requirement under MDR for medical devices that are composed of substances or combinations of substances

How to achieve compliance in the USA? Dietary supplement labels

Probiotics: official definition, scientific requirements and vigilance points during product development

How to achieve compliance in the USA? Structure Function Claims

RNI Infographic – Comparing regulations and requirements of cosmetic products in EU / USA

Cannabidiol novel food evaluations on hold pending new data
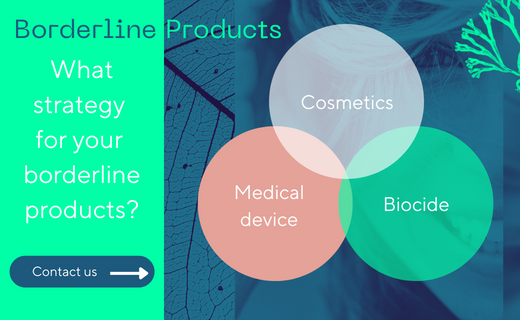
Borderline products

Reg&Co – Regulatory and Design Solution

How to get compliant in the USA? Dietary ingredients

Mutual Recognition Regulation Explained

RNI is committed and supports the associations of the Club House and the SPIO !

Business After Brexit – How can we help?

RNI has forever been evolving with you…

EC Evaluation of Botanical Health Claims and Nutrient Profiles

Face Masks and PPE in the UK during Covid-19

The Impact of Covid-19 on the Regulatory Framework – Part 4

The Impact of Covid-19 on the Regulatory Framework – Part 3

COVID-19: UK’s Regulatory Response to Medical Devices

The Impact of Covid-19 on the Regulatory Framework – Part 1

Hand Gels and Sanitisers – The Grey Zone for Classification

COVID-19: EU Regulatory Response to Disinfectant Products

Key Regulatory Timeline | Nutrition Products & Health Products

Health Claims for Food Supplements in Europe – A Competitive Advantage?
Could Medical Device Regulation be delayed due to Covid-19?

Medical Devices – EUDAMED and UDI’s explained!
1 YEAR MDR DELAY IS POLITICALLY A DONE DEAL!

Indication of the origin of the primary ingredients: What requirements?

Essential oils: a booming industry with an undefined regulatory framework

EU Cosmetic Allergen Labelling: Is online labelling the future?

Class I Medical Device Transition period…

Brexit Day: What comes next?

BREXIT: UK Medicines and Medical Devices Bill

The Impact of Covid-19 on the Regulatory Framework – Part 2

The Probiotic Series – Your e-Book

THE PROBIOTIC SERIES – What Information is Required for a Probiotic GRAS or NDIN Dossier?

Fair trade claims

THE PROBIOTIC SERIES – Determining the Best Pathway to Regulatory Compliance

Cosmetic Regulations Post Brexit : how to prepare for a ‘no deal’ Brexit

THE PROBIOTIC SERIES – Probiotic Labeling

THE PROBIOTIC SERIES – Probiotic Taxonomy

Is your Business Brexit Ready?

Meet RNI at CPhI Europe |November 5 to 7 |Frankfurt

RNI Consulting at Supply Side West (LA) | October 15-19, 2019

Meet RNI Consulting LLC at Supply Side West

RNI Consulting Ltd |Associate Membership PAGB | UK

Conference RNI Conseil | AromaDays |July 4 | Avignon (France)

RNI Consulting Ltd |New Office in London (UK)

CPhI CHICAGO | April 30 to May 2, 2019 | Come and meet us !

Seminaire RNI | Actualités Réglementaires Europe/USA/UK


